Experimentalphysik IV
News
Emmy Noether-Nachwuchsgruppe für Dr. Aisha Aqeel
Dr. Aisha Aqeel wurde mit einer Emmy Noether-Nachwuchsgruppe von der Deutschen Forschungsgemeinschaft (DFG) ausgezeichnet, um eine Forschungsgruppe „Spintronik mit chiralen helimagnetischen Isolatoren“ an der Universität Augsburg aufzubauen. Das Forschungsteam um Dr. Aqeel wird sich der Weiterentwicklung unkonventioneller magnetischer Systeme, so genannter Helimagnete, widmen.
[Universität Augsburg]
Skyrmionen und Antiskyrmionen hoher Ordnung entdeckt
Forschende der Universität Augsburg und der Universität Wien haben in magnetischen Co/Ni-Mehrschichtfilmen bei Raumtemperatur nebeneinander existierende magnetische Skyrmionen und Antiskyrmionen beliebiger topologischer Ladung entdeckt. Ihre Ergebnisse wurden in der renommierten Fachzeitschrift Nature Physics veröffentlicht. Die Entdeckung eröffnet neue Perspektiven in der Skyrmionenforschung.
[Universität Augsburg]
Wie viel Karat gibt eine Kuh?
Den Zusammenhang zwischen Kuh und Diamant gibt es wirklich! Herausgefunden haben das Forschende an der Universität Augsburg um Dr. Matthias Schreck, Arbeitsgruppenleiter Diamant in der Experimentalphysik IV am Institut für Physik der Mathematisch-Naturwissenschaftlich-Technischen Fakultät.
[50 Jahre – viele Geschichten]
Entdeckung: neue magnetische Wirbel für die Datenspeicherung
Skyrmionen sind winzige magnetische Wirbel, sehr stabil und finden zum Beispiel in neuartigen Datenspeichern Anwendung. Forschenden am Lehrstuhl für Experimentalphysik IV ist es nun gelungen, eine neue Art zu nachzuweisen: Antiskyrmionen erster und zweiter Ordnung. Gemeinsam mit Kollegen und Kolleginnen der Universität Wien entwickelten sie das „Rezept“ für die neuen Skyrmionen und publizierten ihre Forschungsergebnisse in Nature Communications.
[Universität Augsburg]
Neue Methode zur Erzeugung von Terahertz-Strahlung
Physiker der Universitäten Augsburg und Münster haben einen neuartigen Emitter zur Erzeugung von Terahertz-Strahlung vorgestellt, der sich durch Variation der Temperatur an- oder abschalten lässt. In Zukunft könnte sie möglicherweise den Bau von Strahlenquellen höherer Intensität ermöglichen. Mit Terahertz-Strahlung lassen sich beispielsweise Fehler in Materialien sichtbar machen oder Hautkrebszellen aufspüren.
[Universität Augsburg]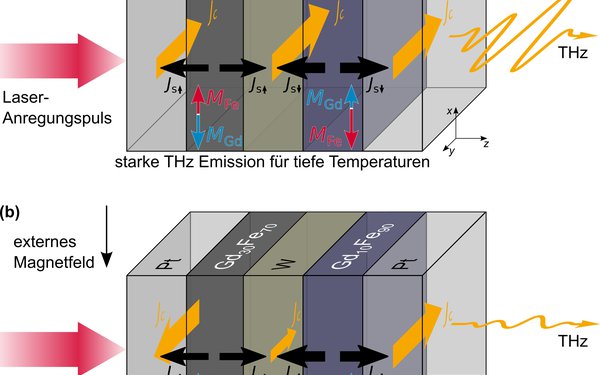
DPG Poster Award 2019
Group Member Alexander Hofmann has won this year's CPP Poster Award with his work on Dipolar doping of organic semiconductors to enhance carrier injection
[Experimentalphysik IV]
Contact information:
Address:
Experimentalphysik IV
Institut für Physik
Universität Augsburg
Universitätsstr. 1
86159 Augsburg
Phone: +49 (0) 821 598-3402 (Office)
Fax: +49 (0) 821 598-3425
E-Mail: sekretariat_ep4@physik.uni-augsburg.de (Office)
Building: R


